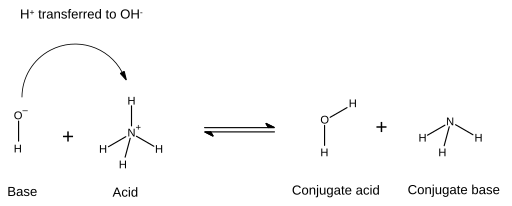
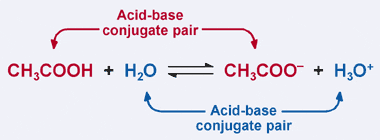
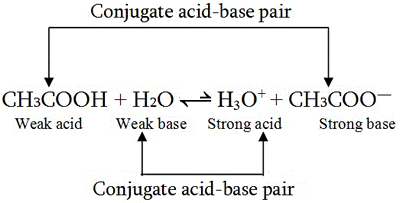
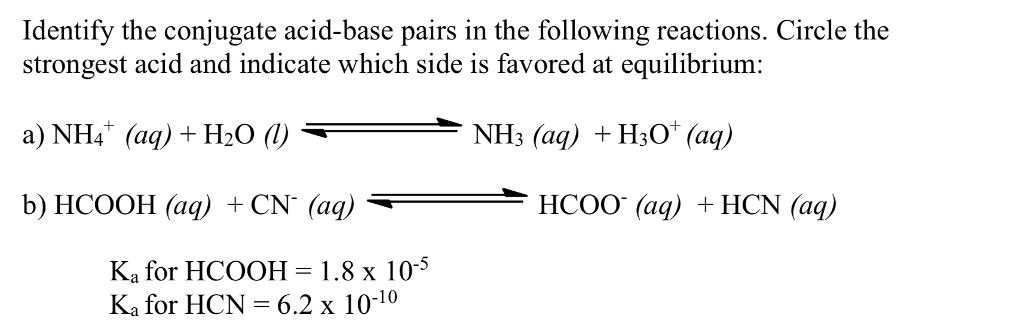Relative strengths of some common conjugate acid- base pairs, listed... | Download Scientific Diagram

20200220 175328 - Conjugate Acid Base Pairs Chem Worksheet 19-2 An acid is defined as a proton H* donor while a base is a proton acceptor. The | Course Hero

SOLVED: Consider the reaction below. Which species are conjugate acid/base pairs? HSO:- (aq) HCN (aq) = HzSOz (aq) + CN- (aq) A) HSOs , CN- B) HSO: , HzSOz C) HzSO3, CN

Relative strengths of some common conjugate acid- base pairs, listed... | Download Scientific Diagram

Identify the conjugate acid-base pairs for the reaction (with the acid written first). CN- + H2O = HCN + OH- |CN- / HCN |HCN / CN- |OH- / H2O |H2O / OH- | Homework.Study.com

Identify the conjugate acid-base pairs in the following reaction. Indicate what each substance is in each pair. H2CO3 + PO43- arrow HCO3- + HPO42- | Homework.Study.com