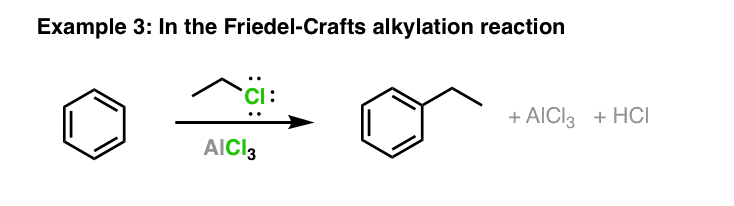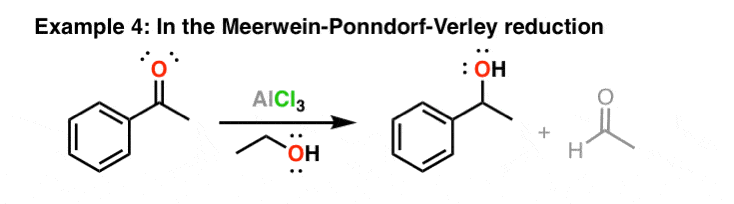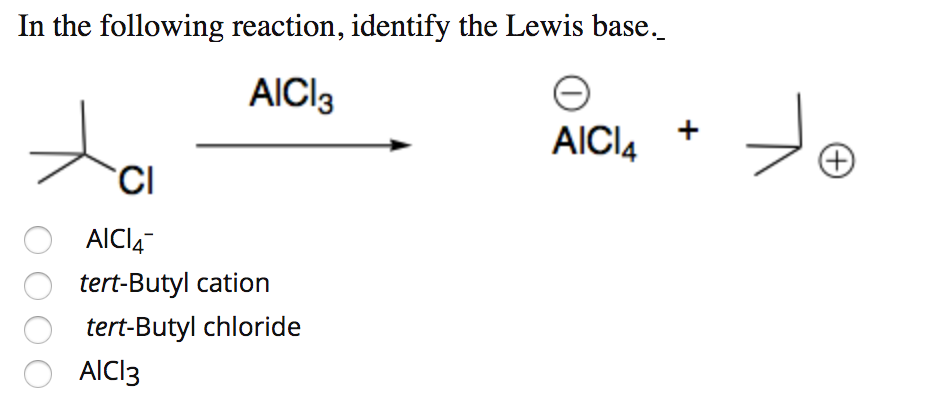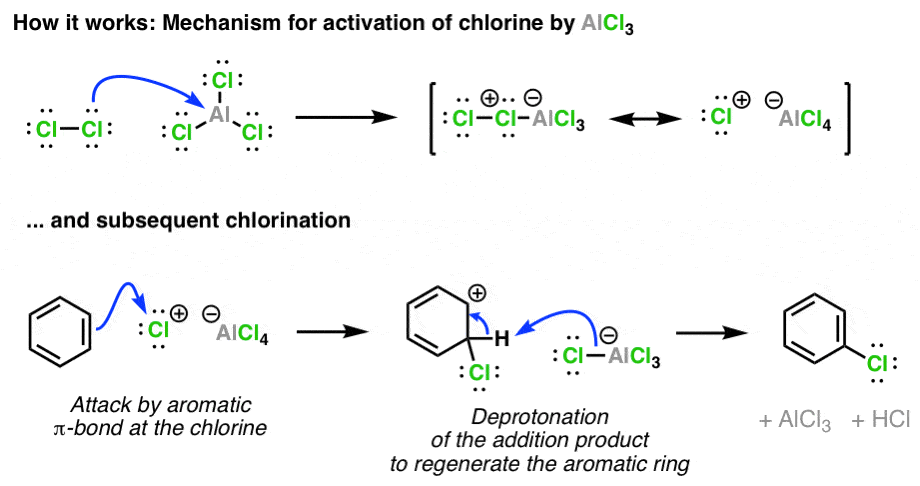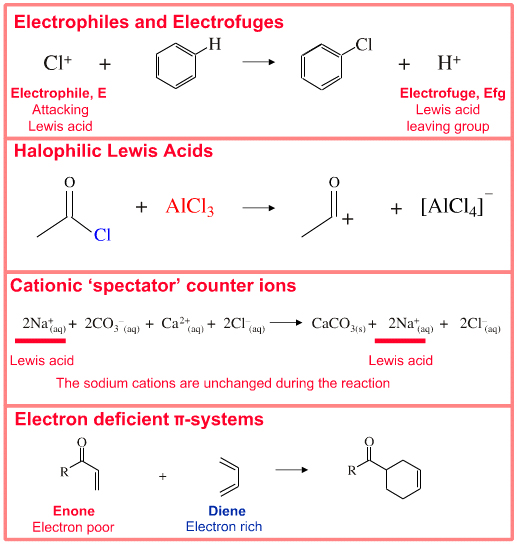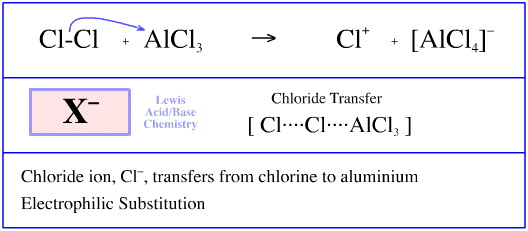
Is the following acid-base reaction Arrhenius, Bronsted-Lowry, or Lewis: AlCl3 + Cl --> AlCl4- | Socratic
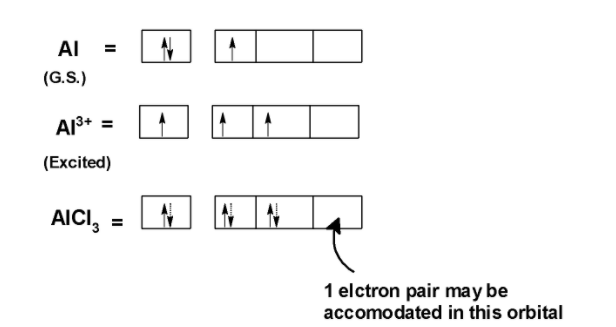
Statement: Aluminium chloride $\\text{ AlC}{{\\text{l}}_{\\text{3}}}\\text{ }$ is a Lewis acid because it can donate the electron.If a given statement is true enter 1 if false enter 0.

Draw the Lewis Structure of the product formed from the reaction between ammonia and aluminum chloride. | Homework.Study.com

AQA A Level chemistry - A2 Unit 5: Section 3.5.5 Reactions of Inorganic Compounds in Aqueous Solution - Lewis acids and bases

Selective synthesis of spirobiindanes, alkenyl chlorides, and monofluoroalkenes from unactivated gem-difluoroalkanes controlled by aluminum-based Lewis acids | Scientific Reports