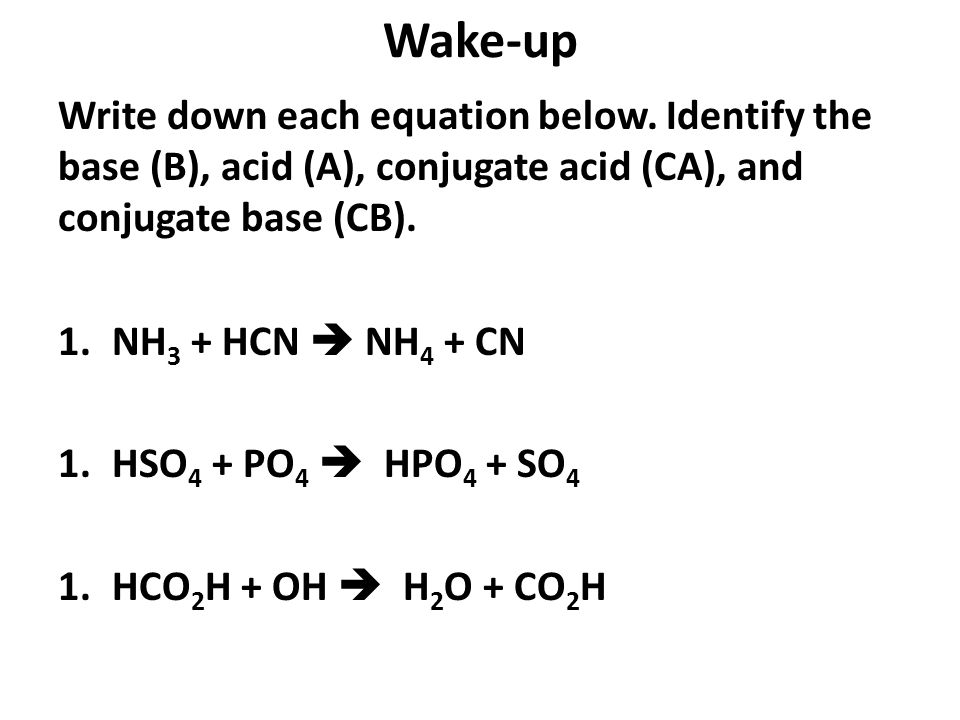
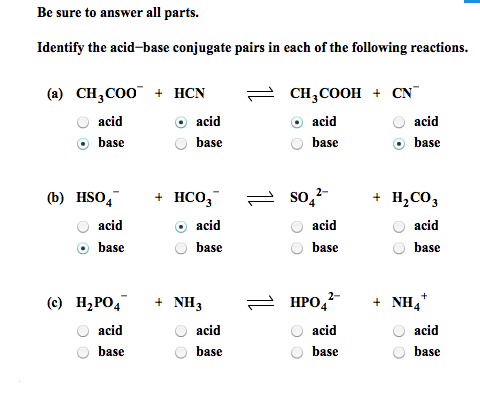SOLVED: The cyanide ion is the conjugate base of the weak acid hydrocyanic acid. The value of Kb for CN-, is 2.50×10-5. Write the equation for the reaction that goes with this

Pd-Catalyzed C–N Coupling Reactions Facilitated by Organic Bases: Mechanistic Investigation Leads to Enhanced Reactivity in the Arylation of Weakly Binding Amines | ACS Catalysis

SOLVED: 4) Fill out the conjugate acids/bases in the following table: Conjugate Acid Base Pairs Acid: HF Conjugate base: Base: SOa" Conjugate acid: Base: Cl Conjugate acid: Acid: HCIOa Conjugate base: Acid: